|
10/4/2023 0 Comments Peptide backbone nitrogen atom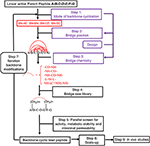
Zwitterionic structure of both proline enantiomers: ( S)-proline (left) and ( R)-proline Biological activity This can then either spontaneously cyclize to form 1-pyrroline-5-carboxylic acid, which is reduced to proline by pyrroline-5-carboxylate reductase (using NADH or NADPH), or turned into ornithine by ornithine aminotransferase, followed by cyclisation by ornithine cyclodeaminase to form proline. Glutamate-5-semialdehyde is first formed by glutamate 5-kinase (ATP-dependent) and glutamate-5-semialdehyde dehydrogenase (which requires NADH or NADPH). Proline is biosynthetically derived from the amino acid L- glutamate. The name proline comes from pyrrolidine, one of its constituents. The next year, Emil Fischer isolated proline from casein and the decomposition products of γ-phthalimido-propylmalonic ester, and published the synthesis of proline from phthalimide propylmalonic ester. Proline was first isolated in 1900 by Richard Willstätter who obtained the amino acid while studying N-methylproline, and synthesized proline by the reaction of sodium salt of diethyl malonate with 1,3-dibromopropane. Proline is the only proteinogenic secondary amino acid which is a secondary amine, as the nitrogen atom is attached both to the α-carbon and to a chain of three carbons that together form a five-membered ring. It is encoded by all the codons starting with CC (CCU, CCC, CCA, and CCG). It is non-essential in humans, meaning the body can synthesize it from the non-essential amino acid L- glutamate. The "side chain" from the α carbon connects to the nitrogen forming a pyrrolidine loop, classifying it as a aliphatic amino acid. The secondary amine nitrogen is in the protonated form (NH 2 +) under biological conditions, while the carboxyl group is in the deprotonated −COO − form. Also note the first peptide chain possesses an internal loop.Proline (symbol Pro or P) is an organic acid classed as a proteinogenic amino acid (used in the biosynthesis of proteins), although it does not contain the amino group -NHĢ but is rather a secondary amine. He found the primary structure to comprise of two chains linked by two cysteine disulfide bridges. This pioneering work, completed in 1953 after some 10 years of effort, earned a Nobel Prize for British biochemist Frederick Sanger (born 1918). Insulin was the first protein whose amino acid sequence was determined. 
Remember that reduction is the addition of hydrogen.Ĭysteine residues in the the peptide chain can form a loop buy forming the disulfide bond (-S-S-), while cysteine residues in different peptide chains can actually link what were otherwise separate chains. The reduction of a disulfide bond is the opposite reaction which again leads to two separate cysteine molecules. This is an important bond to recognize in protein tertiary structure. The sulfurs (yellow) join to make the disulfide bridge. An unspecified oxidizing agent (O) provides an oxygen which reacts with the hydrogen (red) on the -SH group to form water. The oxidation of two cysteine amino acids is shown in the graphic. The oxidation of two sulfhydryl groups results in the formation of a disulfide bond by the removal of two hydrogens. The amino acid cysteine undergoes oxidation and reduction reactions involving the -SH (sulfhydryl group). An important resonance contributor has a C=N double bond and a C-O single bond, with a separation of charge between the oxygen and the nitrogen.Īlthough B is a minor contributor due to the separation of charges, it is still very relevant in terms of peptide and protein structure – our proteins would simply not fold up properly if there was free rotation about the peptide C-N bond.ĭisulfide Bridges and Oxidation-Reduction This, along with the observation that the bonding around the peptide nitrogen has trigonal planar geometry, strongly suggests that the nitrogen is sp 2-hybridized. One of the most important examples of amide groups in nature is the ‘peptide bond’ that links amino acids to form polypeptides and proteins.Ĭritical to the structure of proteins is the fact that, although it is conventionally drawn as a single bond, the C-N bond in a peptide linkage has a significant barrier to rotation, almost as if it were a double bond. Resonance contributors for the peptide bondsĪ consideration of resonance contributors is crucial to any discussion of the amide functional group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
